Martensite and Bainite in the CGHAZ of HSLA Steel Welds
Three forms of bainite are commonly found in high-strength low-alloy (HSLA) steels: upper bainite (B-II), lower bainite (B-III), and granular bainite (B-IV). Both upper bainite and lower bainite (Figures 1 and 2) have ferrite laths, while granular bainite is lath-free and contains martensitic/austenitic (M/A) islands (Figure 3). Lower bainite is similar in appearance to martensite (Figure 4) because its lattice is formed by a similar Bain strain and Kurdjumov-Sachs (K-S) orientation.

Figure 1 Upper bainite [1].
Figure 2 Lower bainite [2].
Figure 3 Granular bainite [3].
Figure 4 Martensite [4].
High cooling rates result in an austenite-to-martensite transformation that is controlled by an instantaneous shear of the austenite lattice to a body centered tetragonal (BCT) lattice where carbon is in solution. This is a high-energy, non-equilibrium, diffusion-less, displacive deformation transformation following the Kurdjumov-Sachs (K-S) orientation:
{111}A//{101}M
<110>A//<111>M
During the formation of bainite, the transformation is time-dependent, and features a diffusion-control mechanism as well as an associated displacive deformation. This results in the formation of a body-centered cubical (BCC) lattice.
Both microstructures are effective in strengthening steels; however, untempered martensite and upper bainite have a negative impact on toughness. By comparison, tempered lath martensite and tempered bainite (Figures 5 and 6) positively impact toughness. Due to their common shearing mechanisms, the lath orientations of martensite and bainite appear similar under a light microscope, making it hard to differentiate the two. This is also true of tempered martensite and degenerated upper bainite (Figure 7).
Figure 5 Tempered martensite [5].
Figure 6 Tempered bainite [6].
Figure 7 Co-existence of martensite and bainite [5]
During continuous cooling of the coarse-grained heat-affected zone (CGHAZ), the phase transformation sequence associated with austenite decomposition is as follows:
austenite → upper bainite → lower bainite → martensite
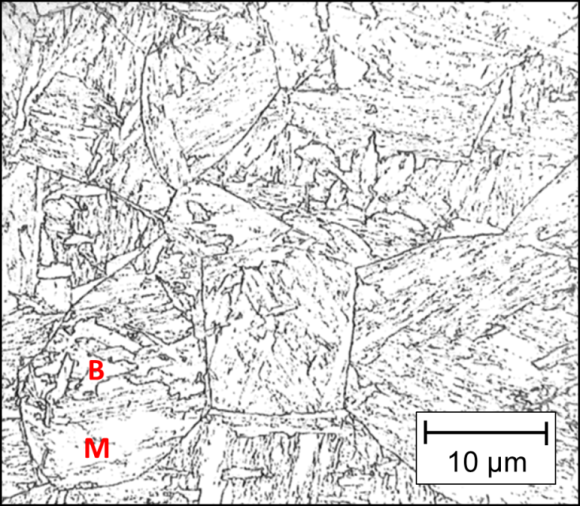
This transformation sequence doesn’t always occur at low heat inputs due to the combination of high cooling rates and chemical undercooling caused by variations in the bulk chemical composition. For example, the banding commonly found in rolled steels may be enriched with carbon (C) and manganese (Mn). C and Mn are known to expand the austenite zone in the Fe-C phase diagram, decreasing the austenite decomposition temperature. If this temperature is close to the martensite start temperature, the diffusion-controlled phase transformation necessary to form upper bainite will be limited. This undercooling simultaneously promotes the formation of lower bainite and martensite. The high cooling rate of low heat-input welds further reduces the time for diffusion to occur, resulting in the formation of lower bainite + lath martensite in the CGHAZ (Figure 8).
Figure 8 Bainite + martensite mix structure [7]
When HSLA steels are welded using high heat inputs, the resultant slow cooling rate elongates the time for the transformation from austenite to upper bainite, which leads to the carbon partitioning that promotes the degeneration of ferrite laths (Figure 7). As a result, the structure is difficult to distinguish from tempered martensite (Figure 6 and 7).
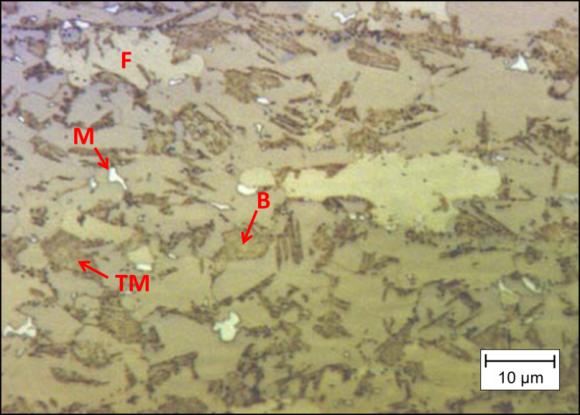
Lepera’s etchant can be used on steels to bring about a sharp contrast between the bainite (brown color), fresh martensite (white color) and M/A constituents (black color) (Figure 9). However, it is still difficult to differentiate tempered martensite (dark brown color) from bainite due to their color similarity.
Figure 9 Use of Lepera’s etchant to delineate microstructure.
References
- Used with permission. ASM Handbook, 2004, Vol. 9, “Metallography and Microstructure”, p182.
- Used with permission. Peet, M.J. 07/2010, “Transformation and tempering of low-temperature bainite”.
- Used with permission. ASM Handbook, 2004, Vol. 9, “Metallography and Microstructure”, p614.
- Used with permission. ASM Handbook, 2004, Vol. 9, “Metallography and Microstructure”, p170.
- Used with permission. ASM Handbook, 2004, Vol. 9, “Metallography and Microstructure”, p169.
- Used with permission. ASM Handbook, 2004, Vol. 9, “Metallography and Microstructure”, p613.
- Used with permission. X. Yue, et at., 1012, “Continuous Cooling Transformation Behavior in the CGHAZ of Naval Steels”, Welding Journal, Vol. 91, No.3, p67-s to 75-s.